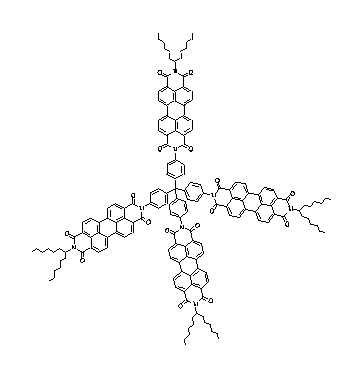
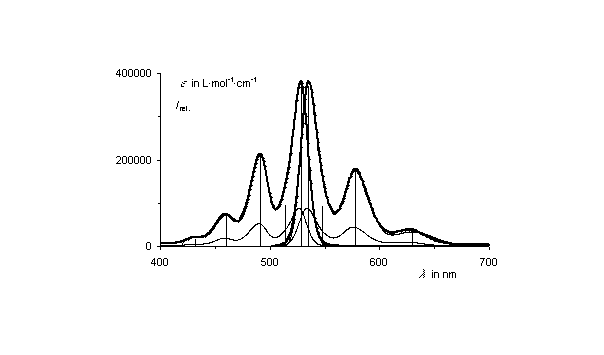
A complete gaussian analysis of the UV/Vis absorption and fluorescence spectrum of the star-like oriented multichromophoric perylene fluorescent systems was successful.
The main topic of the research activities has been the
chemistry and physics of highly photostable fluorescent dyes.
Preparation procedures for novel fluorescent dyes were developed
and the properties of the dyes investigated. Several novel
multichromophoric dyes were synthesized; such substances are for
example of special interest because natural photosynthesis
reaction centers consist of similar assemblies and form the basis
for the collection of solar energy for plants and some bacteria.
The interaction of the chromophores in these systems were studied
on the basis of a lineshape analysis of their UV/Vis spectra.
Gaussian functions proved to be the more appropriate description
for the lineshape of the UV/Vis spectra, whereas the also
frequently used log normal function gave appreciably poorer
results; this was especially shown for structured UV/Vis spectra.
The novel dyes can be used as fluorescent standards because of
their unusually good properties such as high fluorescence quantum
yield and photostability that exceed the known fluorescent
standards appreciably. Furthermore, the novel fluorescent dyes
are of interest for dye laser applications and for light
collecting assemblies. Several novel analysis methods could be
developed, for example for the detection of aldehydes by membrane
detectors and for the immunochemical determination of traces or
the development of receptor tests. The latter are suitable for
the detection of traces of pollutants in the environment. Dyes
with universal anchor groups have been developed.
Highly photostable fluorescent dyes are useful for optical
recording systems; the latter are going to be further developed.
Beyond these dye specific activities, various reaction mechanistic problems were investigated, for example concerning the quantification of solvent effects in binary liquid mixtures and mechanisms of rearrangement reactions.
Further subjects concern polymer chemistry, for example the structure of humic substances.
The tetrahedral arrangement of perylene bisimide chromophores gives a novel molecular system of antennae for the conversion of diffuse solar radiation. A detailed analysis of their UV/Vis spectra gives an impression of the consolidation of the single chromophores.


A complete gaussian analysis of the UV/Vis absorption and
fluorescence spectrum of the star-like oriented multichromophoric
perylene fluorescent systems was successful.
·H. Langhals, C. Wagner, R. Ismael, New. Journ. Chem. 2001, 25, 1047-1049.
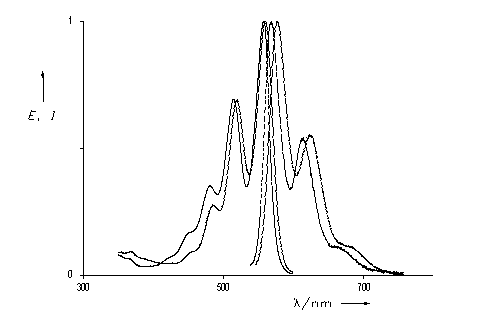
The light harvesting unit B800 of the bacteria system LH2 has been simulated by the ring-type arrangement of up to six benzoperylene trisimide chromophores. These multichromophoric arrangements are highly fluorescent. A Gaussian analysis was successful for their UV/Vis spectra. Exciton coupling of the single chromophores was found in this arrangement and was discussed.
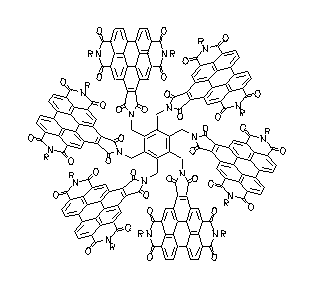
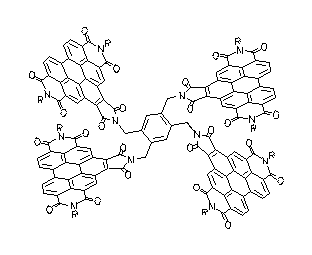
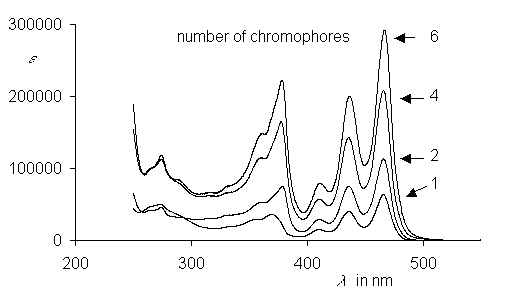
UV/Vis absorption spectra of ring-type multichromophoric
benzoperylene fluorescence dyes. From top to bottom:
six, four, two and one chromophores.
·H. Langhals, M. Speckbacher, Eur. J. Org. Chem. 2001, 2481-2486.
The light-fast bichromophoric dye C25 is distinguished
by 100% energy transfer from the benzoperylene trisimide to the
perylene bisimide chromophor and exhibits a fluorescence quantum
yield of 100%, even in the presence of atmospheric oxygen. The
absorption spectrum of C25 consists of many bands for
fluorescence excitation because of the two chromophores. The
fluorescence quantum yield is consistently high over the whole
visible region and this can be even extended to the UV.
C25 can be therefore used as a standard for the
calibration of fluorescence spectrometers.

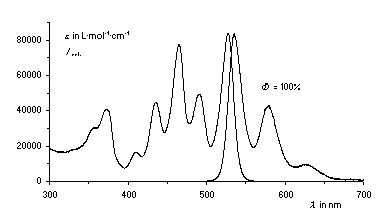
UV/Vis-absorption and fluorescenece spectra of the dye C25.
·S. Kalinin, M. Speckbacher, H. Langhals, L. B.-Å. Johansson, Phys. Chem. Chem. Phys. 2001, 3, 172-174.
Aggregation is the most important reason for the quenching of
fluorescence at high concentrations of dyes.
Such an aggregation can be effectively suppressed by the
attachement of bulky aryl or highly branched alkyl groups to the
nitrogen atoms of perylene dyes. The dyes thus obtained exhibit
strong fluorescence even in highly concentrated solutions or in
the solid-state.

·H. Langhals, R. Ismael, O. Yürük, Tetrahedron 2000, 56, 5435-5441.
The log normal function and the Gaussian function are used to equal extents in the literature for the description of the lineshape of UV/Vis spectra in solution. A comparison of both functions with experimental data is still lacking.
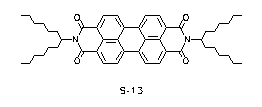
We have fitted both functions to the strongly structured UV/Vis spectrum of perylene dye S-13 and obtained an appreciably better result (R = 1.7%) for the Gaussian function (e = Semax·exp (- (1/l - 1/lmax)2/(2·s2))) compared to log normal functions. A complete Gaussian analysis both for the absorption and fluorescence spectra of S-13 was successful. The distribution of intensities of the vibronic bands could be described with the Ross function.
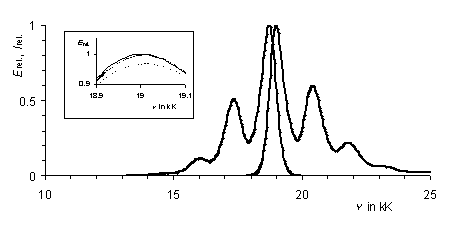
UV/Vis absorption and fluorescence spectra of S-13 in
chloroform (thick line). Simulated spectra by the Gaussian
function (thin line). Insert: from top to bottom: experimental
spectrum, best fit for Gaussian function and log normal function
(dotted line).
·H. Langhals, Spectrochim. Acta Part A, 2000, 56, 2207-2210.
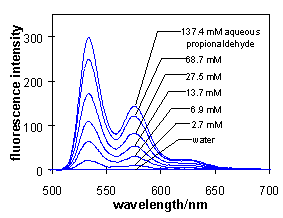
The fluorescence of N-aminoperylene-tetracarboxylic
bisimides is quenched by an intramolecular electron transfer
process from the amino group to the electronically excited
aromatic system. This type of quenching of fluorescence will be
switched off by the condensation of the amino group with
aldehydes and the derivatives thus obtained exhibit strong
fluorescence. This is applied in a sensitive analysis method for
aldehydes where a hydrophobic aminobisimide is dissolved in a
membrane and brought into contact with an aqueous solution of the
aldehyde. The aldehyde induces reversibly a fluorescence of the
membrane for its continous detection.
A typical time of a reversible response of 2...7 min was observed
for such a detector for a step-wise alteration of the
concentration by a factor of 10 (t95) in both
directions at a pH of 2.5. A typical dynamic region of 5 ... 100
mmol/L and a detection limit of 0.08 mmol/L was obtained.

 Membrane fluorescence
induced |
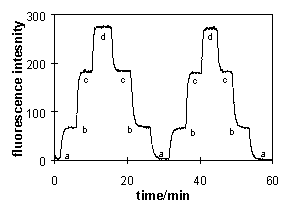 Reversibility of the
induced |
·G. J. Mohr, U. E. Spichiger, W. Jona, H. Langhals, Anal. Chem. 2000, 72, 1084-1087.
Perylene-3:4,9-tricarboxylic imides are prepared in a one-step synthesis from perylene-3,4:9,10-tetracarboxylic-3,4-anhydride-9,10-imides by partial decarboxylation with copper powder. The imides are of interest as labels because of their intense fluorescence and light-fastness and their easy monofunctionalization. A derivatization of amines and alcohols with these compounds is demonstrated.
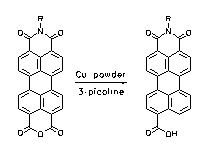
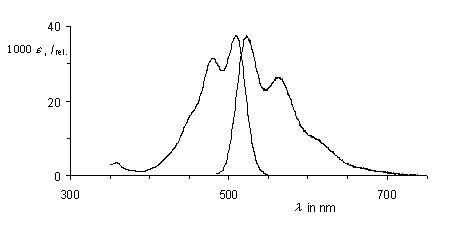
UV/Vis absorption and fluorescence spectra of
perylenetricarboxylic imides.
·F. Süßmeier, H. Langhals, Eur. J. Org. Chem. 2001, 607-610.
Core-extended perylene tetracarboxylic bisimides were prepared by Diels-Alder reaction or by nitration followed by reduction and cyclisation, respectively. Highly fluorescent dyes were obtained with absorption regions from the visible to the NIR. Applications, for example, for solar energy harvesting, and quantum counters were suggested.
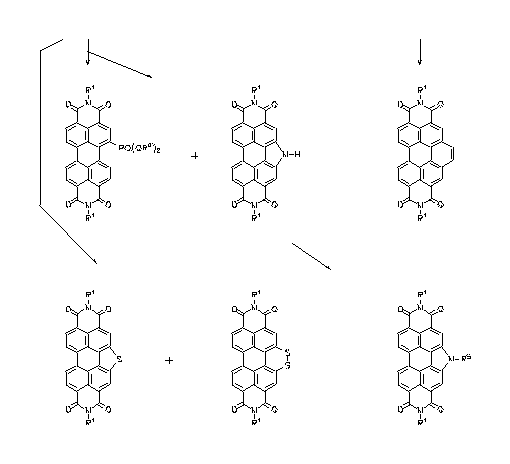
UV/Vis-absorption, fluorescence and solid-state fluorescence
spectra
of benzoperylene trisimides
·H. Langhals, S. Kirner, Eur. J. Org. Chem. 2000, 365-380.
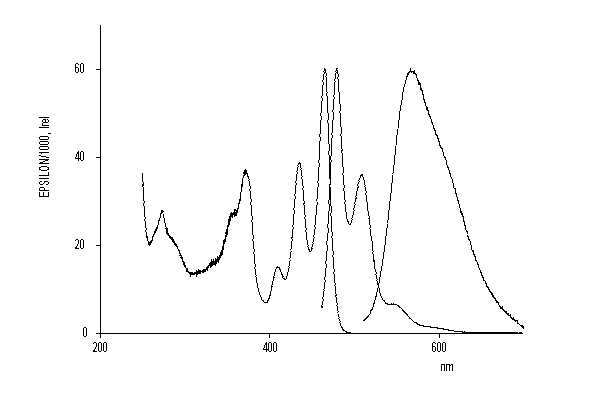
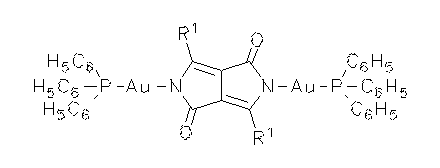
DPP-type dye pigments were deprotonated and coordinated with transition metals. Dyes with novel properties were thus obtained such as high solubilities and high fluorescence quantum yields. X-ray crystal structure analysis indicate twists of the planes of the substituents R1 versus the planes of chromophore depending on the complex fragment.
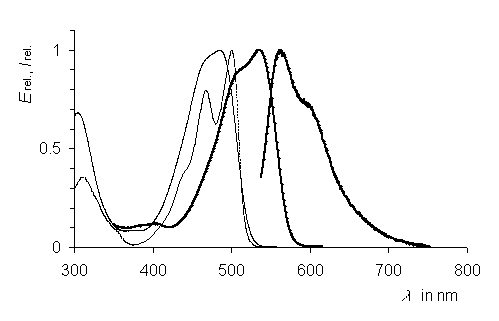
UV/Vis absorption and fluorescence spectra of the
DPP gold complex (thick line) compared to
unsubstituted DPP (thin line right) and the
N-methyl DPP (thin line left).
·H. Langhals, M. Limmert, I.-P. Lorenz, P. Mayer, H. Piotrowski, K. Polborn, Eur. J. Inorg. Chem. 2000, 2345-2349.
Perylene imides 2 were presented as highly stable fluorescence standards and for laser applications. The UV/Vis absorption spectra of the dyes were adopted to the requirements of information recording by small structural modifications. One of these dyes (4) can replace terrylene (1). This is of special interest for the tunable rhodamine 6G dye laser.
 |
 |
UV/VIS spectra of dye 4 for replacement of terrylene
(spectra of terrylene on the left side).
·H. Langhals, H. Jaschke, P. von Unold, U. Ring, Journ. Inform. Rec. 2000, 25, 195-110.
New perylene fluorescent dyes were applied both in homogeneous solution and as colloidal particles for tracer applications in the investigation of solute and colloidal transport processes in fractured rock. The horizontal and vertical movement of ground water in pores were studied.These effects depend also on the pH value of water.
·IAHS Publ. (Tracers and Modelling in Hydrogeology) 2000, 262, 175-179; Chem Abstr. 2000, 133, 240814.
The UV/Vis-absorption spectra of an aquatic humic system left in its naturally state were measured in the range of concentration of DOC 0.001 ... 1.4 g/l with precision. Lambert-Beers' law is rather well fullfilled at low concentrations whereas deviations have been observed for DOC larger than 0.1 g/l which are interpreted in terms of self solvation and formation of micelles, respectively.
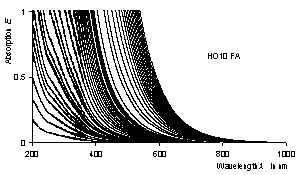 Precision UV/Vis spectra |
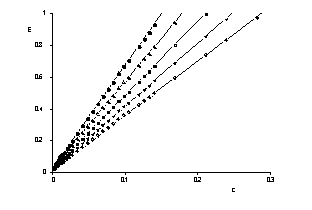 Verification of Lambert Beer's law |
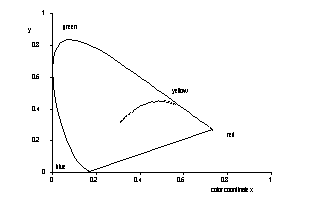 Colour trace of humic substances |
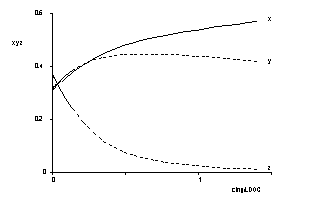 Colour coordinates of humic substances |
·H. Langhals, G. Abbt-Braun, F. H. Frimmel, Acta hydrochim. et hydrobiol. 2000, 28, 329-332.
1. F. Einsiedl, H. Langhals, P. Maloszewski, K. Witthuser, S. Wohnlich, 'Application of two new fluorescent dyes and fluorescent particles in a horizontal and vertical fracture' IAHS Publ. (Tracers and Modelling in Hydrogeology) 2000, 262, 175-179; Chem Abstr. 2000, 133, 240814.
2. H. Langhals, S. Kirner, 'Novel Fluorescent Dyes by the Extension of the Core of Perylene-tetracarboxylic Bisimides', Eur. J. Org. Chem. 2000, 365-380.
3. G. J. Mohr, U. E. Spichiger, W. Jona, H. Langhals, 'Using N-Aminoperylene-3,4:9,10-teracarboxyl-bisimide as a Fluorogenic Reactand in the Optical Sensing of Aqueous Propionaldehyde', Anal. Chem. 2000, 72, 1084-1087.
4. H. Langhals, H. Jaschke, P. von Unold, U. Ring, 'Matched Dyes for Information Recording', Journ. Inform. Rec. 2000, 25, 195-110.
5. H. Langhals, 'A Re-examination of the Line-Shape of the Electronic Spectra of Complex Molecules in Solution. Log-Normal Function Versus Gaussian', Spectrochim. Acta Part A, 2000, 56, 2207-2210.
6. H. Langhals, R. Ismael, O. Yürük, 'Persistent Fluorescence of Perylene Dyes by Steric Inhibition of Aggregation', Tetrahedron 2000, 56, 5435-5441.
7. H. Langhals, M. Limmert, I.-P. Lorenz, P. Mayer, H. Piotrowski, K. Polborn, 'Chromophores Encapsulated in Gold Complexes: DPP Dyes with Novel Properties', Eur. J. Inorg. Chem. 2000, 2345-2349.
8. H. Langhals, G. Abbt-Braun, F. H. Frimmel, 'Association of Humic Substances: Verification of Lambert-Beers Law', Acta hydrochim. et hydrobiol. 2000, 28, 329-332.
9. S. Kalinin, M. Speckbacher, H. Langhals, L. B.-Å. Johansson, 'A New and Versatile Fluorescence Standard for Quantum Yield Determination', Phys. Chem. Chem. Phys. 2001, 3, 172-174.
10. F. Süßmeier, H. Langhals, 'Novel Fluorescence Labels: the Synthesis of Perylene-3:4,9-tricarboxylic Imides', Eur. J. Org. Chem., 2001, 607-610.
11. H. Langhals, M. Speckbacher, 'Intramolecular Effects in Covalently Connected Units: Ring-type Oriented Chromophores and their Interactions', Eur. J. Org. Chem. 2001, 2481-2486.
12. H. Langhals, C. Wagner, R. Ismael, 'Star-like oriented chromophores', New. Journ. Chem., 2001, 25, 1047-1049.
1. H. Langhals, R. Ismael, O. Yürük, 'Perylenfarbstoffe mit persistenter Fluoreszenz durch sterische Aggregationshinderung', Ger. Offen. DE 10026623.1 ( May 29, 2000).
2. H. Langhals, F. Süßmeier, 'Neue Fluoreszenzlabels: die Synthese von Perylen-3:4,9-tricarbonsäureimiden',Ger. Offen. DE 10038672.5 (August 8, 2000).
3. H. Langhals, 'Kompetitive Prionen-Reagenzien und ihre Anwendung in Diagnostik und Therapie', Ger. Offen. DE 10104279.5 (January 31, 2001).
4. H. Langhals, 'Farbstoffe mit sternförmig angeordneten Chromophoren, ihre Synthese, spektralen Besonderheiten und Verwendung', Ger. Offen. DE 10105238.3 (February 6, 2001).
5. H. Langhals, P. Blanke, 'Neue NIR-Farbstoffe auf Perylen-Basis', Ger. Offen. DE 10132116.3 (July 3, 2001).
1. Presentation at the trade-faire Hannovermesse, 'High performance fluorescent dyes', Hannover, April 23 ... April 28, 2001.
2. Presentation at the trade-faire Laser 2001, 'High performance fluorescent dyes', Munich, June 18 ... June 22, 2001.
H. Langhals, 2009. - Impressum - Datenschutz - Kontakt