

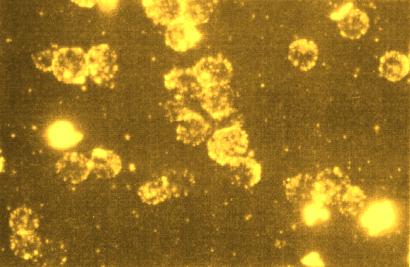
Shining points: fluorescent immunoliposomes. They are mainly bound to cells.
The lipid double layer of liposomes was doped with the perylene dye S-17 and linked to antibodies. The highly fluorescent antibodies thus labelled could be detected with a simple fluorescence microscope and visually traced to their binding site. The labelled antibodies could be stored for more than one year. There are many of different applications of such labelled antibodies in analytics and diagnostics.
|
 Shining points: fluorescent immunoliposomes. They are mainly bound to cells. |
· H. Langhals in B. Hock, Immunochemical Detection of Pesticides and their Metabolites in the Water Cycle, VCH Verlagsgesellschaft, Weinheim 1995, ISBN 3-527-27137-6; Chem. Abstr. 1996, 124, 24966z. - H. Schott, D. v. Cunov, H. Langhals, Biochim. Biophys. Acta 1992, 1110, 151-157. - R. A. Schwendener, T. Trüb, H. Schott, H. Langhals, R. F. Barth, P. Groscurth, H. Hengartner, Biochim. Biophys. Acta 1990, 1026, 69-79.
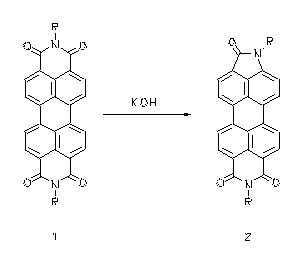
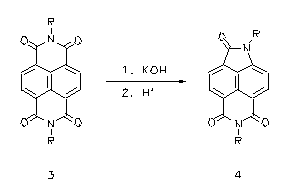
Tetracarboxylic bisimide lactame ring contractions - a novel type of rearrangement
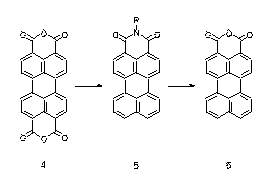
The reaction of alcoholic alkali with aromatic tetracarboxylic bisimides such as perylene tetracarboxylic bisimides (1) or naphthalene tetracarboxylic bisimides (3) causes their transformation to the corresponding lactame imides (2 and 4, respectively). This transformation is a novel type of rearrangement. The lactame imdides absorb appreciably more bathochromically than the corresponding bisimides. Thus coloured lactamimides (4) were obtained from the colourless naphthalene tetracarboxylic bisimides (3).
 |
 |
||||
|
|
· H. Langhals, P. v. Unold, Angew. Chem. 1995, 107, 2436-2439; Angew. Chem., Int. Ed. Engl. 1995, 34, 2234-2236.
The extraordinarily high stability of cyclic carboxylic imides is reported in a review and demonstrated with the highly stable perylene dyes
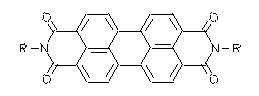
· H. Langhals, Heterocycles 1995, 40, 477-500. - See also: H. Langhals, Chimia 1994, 48, 503-505.
The synthesis of perylene-3,4-dicarboxylic imides
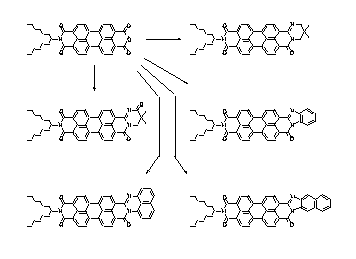
There is no simple and universal synthetic access to perylene-3,4-dicarboxylic imides (5) in contrast to the perylene-3,4:9,10-tetracarboxylic bisimides. These perylene-3,4-dicarboxylic imides (5) could be prepared in a single-step-synthesis from the technical perylene bisanhydride (4) by a water-induced decarboxylizing cyclization. The perylene imides (5) are highly fluorescent and are universal synthetic intermediates, for example, for the synthesis of multichromophoric dyes.
· L. Feiler, H. Langhals, K. Polborn, Liebigs Ann. Chem. 1995, 1229-1244.
Perylene amidinimide dyes
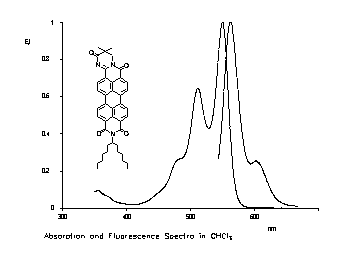
The exchange of one carbonyl group of perylene-3,4-9,10-tetracarboxylic bisimides versus a C=N-unit results in a bathochromic shift of absorption and fluorescence. Thus, highly red to orange fluorescent dyes were obtained.
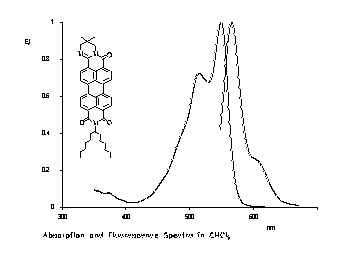 |
 |
||
|
 |
· H. Langhals, S. Sprenger, M.-T. Brandherm, Liebigs Ann. Chem. 1995, 481- 486.
The determination of molecular weights of dyes by GPC
It could be demonstrated with perylene dyes that gel permeation chromatography is suitable for the determination of the molecular weights of dissolved dyes. This is of interest because of the low voltailibility of many dyes and is of special importance for the analysis of bi- tri- and multichromophoric dyes.
· H. Langhals, R. Kollefrath, J. Lindner, Macromol. Rep. 1995, A32, 415-423.
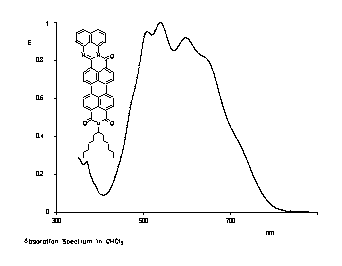
Quaterrylene tetracarboxylic bisimides
Quaterrylene tetracarboxylic bisimides were prepared in a two-step synthesis and are of interest as NIR fluorescent dyes.

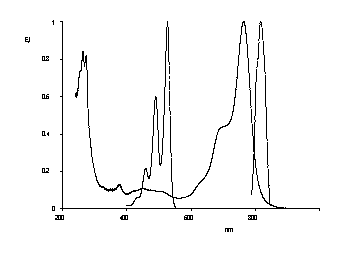
UV/Vis absorption and fluorescence spectra of
quaterrylene bisimides compared with the
absorption spectrum of perylene bisimides.
· H. Langhals, G. Schönmann, L. Feiler, Tetrahedron Lett. 1995, 36, 6423-6424.
1,7-Diazaperylene
The photophysical behaviour of the novel heterocycle 1,7-diazaperylene (14) in absorption and fluorescence was investigated.
 |
 |
 |
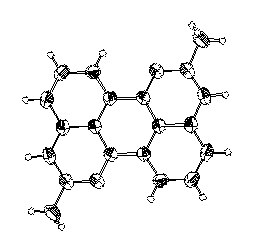 |
· T. Sander, H.-G. Loehmannsroeben, H. Langhals, J. Photochem, Photobiol. A 1995, 86, 103-108.
DPP-dyes; the influence of asymmetric media in the conformation of diketopyrrolppyrroles
The influence of an anisotropic environment in the conformation of DPP-dyes (15) was investigated by means of polarized light in liquid crystals.

· P. Edman, L. B.-Å. Johansson, H. Langhals, J. Phys. Chem. 1995, 99, 8504-8509.
1. H. Langhals, 'Novel perylene derivatives as highly photostable fluorescent dyes', Chimia 1994, 48, 503-505.
2. H. Langhals, 'Novel dyes for electrophotographic processes with the perylene structure element', 10th IS&T's Int. Congr. Adv. Non-Impact Print. Technol., IS&T-The Society for Imaging Science and Technology, Springfield, Va., 1994; Chem. Abstr. 1996, 124, 59054c.
3. T. Sander, H.-G. Loehmannsroeben, H. Langhals, 'Photophysical and fluorescence quenching properties of 1,7-diazaperylene in solution', J. Photochem, Photobiol. A 1995, 86, 103-108.
4. H. Langhals, S. Sprenger, M.-T. Brandherm, 'Peryleneamidine-imide Dyes', Liebigs Ann. Chem. 1995, 481- 486.
5. P. Edman, L. B.-Å. Johansson, H. Langhals, 'Polarized Light Spectroscopy of Dihydro-pyrroledione in Liquids and Liquid Crystals: Molecular Conformation and Influence by an Anisotropic Environment', J. Phys. Chem. 1995, 99, 8504-8509.
6. L. Feiler, H. Langhals, K. Polborn, 'Synthesis of Perylene-3,4-dicarboximides - Novel, Highly Photostable Fluorescent Dyes', Liebigs Ann. Chem. 1995, 1229-1244.
7. H. Langhals, R. Kollefrath, J. Lindner, 'The determination of the molecular weights of perylene dyes by GPC', Macromol. Rep. 1995, A32, 415-423.
8. H. Langhals, P. v. Unold, 'Tetracarboxylic Bisimide-Lactame-Ring-Contractions - a Novel Type of Rearrangement', Angew. Chem. 1995, 107, 2436-2439; Angew. Chem., Int. Ed. Engl. 1995, 34, 2234-2236.
9. H. Langhals, 'Cyclic carboxylic imide structures as structure elements of high stability. Novel developments in perylene dye chemistry', Heterocycles 1995, 40, 477-500.
10. H. Langhals, G. Schönmann, L. Feiler, 'A Two-Step Synthesis of Quaterrylenetetracarboxylic Bisimides - Novel NIR Fluorescent Dyes', Tetrahedron Lett. 1995, 36, 6423-6424.
11. H. Langhals, 'Dyes for Fluorescent Immunoassays', in B. Hock, Immunochemical Detection of Pesticides and their Metabolites in the Water Cycle, VCH Verlagsgesellschaft, Weinheim 1995, ISBN 3-527-27137-6; Chem. Abstr. 1996, 124, 24966z.
1. H. Langhals, 'Preparation of high-purity perylene-3,4,9,10-tetracarboxylic acid 3,4-anhydride 9,10-imides', Ger. Offen. DE 4421347.6 (June 17, 1994); Chem. Abstr. 1996, 124, 263384q.
2. H. Langhals, 'Perylene dyes with 2 to 4 chromophores, their preparation from formamides and their use', Ger. Offen. DE 4440242.2 (November 10, 1994); Chem. Abstr. 1996, 125, 35829d.
3. H. Langhals, G. Schönmann, 'Fluorescent quaterrylenebisimides, their preparation and use', Ger. Offen. DE 19512773.0 (April 5, 1995); Chem. Abstr. 1996, 125, 303255c.
4. H. Langhals, P. v. Unold, 'Aminonaphthalenetricarboxylic lactam imide fluorescent dyes, their preparation and their use', Ciba Specialty Chemicals Holding Inc., Eur. Pat. Appl. EP 769532 (October 12, 1995); Chem. Abstr. 1997, 127, 6135k.
H. Langhals, 2009. - Impressum - Datenschutz - Kontakt